Project Overview
This project was focused on creating art using the chemistry that we have been learning about. We have been learning about the different types of chemical reactions. For the project, we had to create a piece of art using the chemical reactions that we had learned about in class. The driving question was, "How can we use chemistry to design a work of art for our new STEM building?". We had to pitch our project idea, create the piece of art, then describe the science behind making it. My group decided to bake a minecraft themed cake as our chemical art. We focused on the science of baking, making, decorating, and tasting, with each group member specializing in one of the categories and performing extensive research on it. We were mostly successful in baking our cake, but it did turn out a bit dry.
Main Concepts
Types of Reactions: Our project was focused around baking. This involves numerous chemical reactions, including a decomposition reaction when baking powder is mixed with heat and water.
Combustion Reaction- a substance combines with oxygen, releasing a large amount of energy in the form of light and heat
Synthesis Reaction- two or more substances combine to form a new compound
Decomposition Reaction- A single compound undergoes a reaction that produces two or more simpler substances
Single Replacement Reaction- one element replaces a similar element in a compound
Double Displacement Reaction- the ions of two compounds exchange places in an aqueous solution to form two new compounds
Acid-Base Reaction- an acid and a base react to form water and a salt
Balancing Chemical Reactions: When chemical reactions occur, the equations have to be properly balanced. The first guideline for balancing chemical equations is the law of conservation of mass. The mass, or the number of each atom, of the reactants has to be equal to the mass, or the number of atoms, of the products. The charges also have to be balanced. Each compound has to be correctly balanced so that the charges add up to zero. Al + O2 --> Al2O3
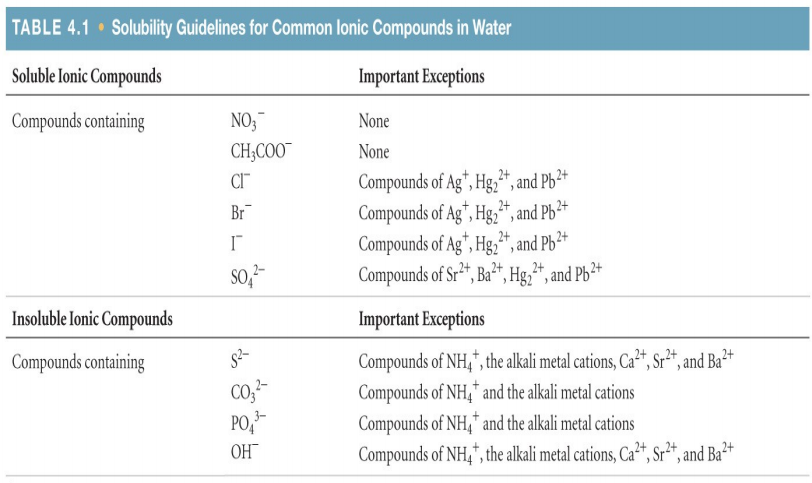
Solubility Guidelines and Reactivity Series:
Solubility is how well a solute dissolves in a solvent. Solubility is affected by temperature, pressure, agitation, the nature of the solute, and the nature of the solvent. Different solutes will dissolve differently in different solvents, which can be determined by the solubility guidelines.
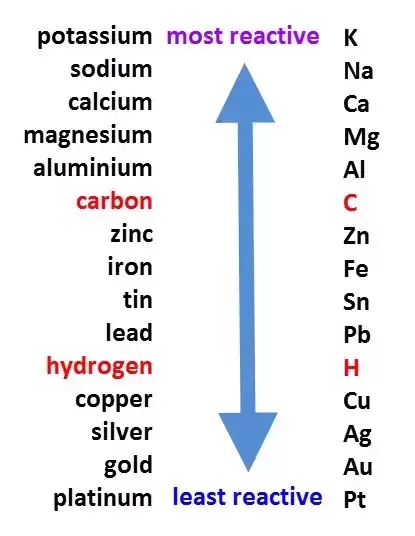
The reactivity series is a list of metals, in order from most to least reactive. It can be used to determine whether or not one metal will replace another in a single replacement reaction. If the first metal is higher than the second in the series, it will replace the second metal in the solution.
Combustion Reaction- a substance combines with oxygen, releasing a large amount of energy in the form of light and heat
Synthesis Reaction- two or more substances combine to form a new compound
Decomposition Reaction- A single compound undergoes a reaction that produces two or more simpler substances
Single Replacement Reaction- one element replaces a similar element in a compound
Double Displacement Reaction- the ions of two compounds exchange places in an aqueous solution to form two new compounds
Acid-Base Reaction- an acid and a base react to form water and a salt
Balancing Chemical Reactions: When chemical reactions occur, the equations have to be properly balanced. The first guideline for balancing chemical equations is the law of conservation of mass. The mass, or the number of each atom, of the reactants has to be equal to the mass, or the number of atoms, of the products. The charges also have to be balanced. Each compound has to be correctly balanced so that the charges add up to zero. Al + O2 --> Al2O3
Solubility Guidelines and Reactivity Series:
Solubility is how well a solute dissolves in a solvent. Solubility is affected by temperature, pressure, agitation, the nature of the solute, and the nature of the solvent. Different solutes will dissolve differently in different solvents, which can be determined by the solubility guidelines.
The reactivity series is a list of metals, in order from most to least reactive. It can be used to determine whether or not one metal will replace another in a single replacement reaction. If the first metal is higher than the second in the series, it will replace the second metal in the solution.
Chemistry of Making |
. Flour and water react to create gluten
. Elastic networks created with gluten . Sifted flour is aerated . Whites (or proteins) create an extra layer around air pockets . Whites coagulate under heat, giving structure . Adds more water . Sugar cuts through butter . Butter forms a layer, creating air pocket . Bicarbonate that reacts with a base . Releases carbon, making cake rise |
Chemistry of Baking |
. Baking powder with heat and water create CO2
. NaHCO3 + H+ → Na+ + H2O + CO2 . Make cake rise . Maillard Reaction: reaction between amino acids, reduces sugars, and heat > creates browning . Not to be mixed up caramelization: oxidation of sugar . Carbonyl group (end with carbon) of sugar + amino acid + heat > N-substituted glycosylamine and water . Glycosylamine undergoes Amadori rearrangement (rearrangement of structure to form a ketose) to form ketosamines . Ketosamines further break down to create a brown color and odor . Functional group: groups of molecules that give its compound unique characteristics . N-substituted glycosylamine: organic sugar compound . Ketosamines: organic compound made of a ketose and amine . Ketose: organic molecule from ketone functional group, monosaccharide . Amine: a compound characterized by the presence of a nitrogen atom |
Chemistry of Taste |
. Sweet taste comes from sucrose- C12H22O11- disaccharide- glucose and fructose
. Sucrose binds to specific membrane receptor proteins in the taste receptor cells (taste buds) . They bind to specific spots on the membrane that only that chemical can bind to because of its shape . A series of chemical reactions are triggered inside the cell, which lead to a change in the flow of ions and the release of a neurotransmitter . In the case of saccharides, G-protein-coupled receptors are activated, which depolarizes taste cells by by activating adenylate cyclase . Then, the cAMP (cyclic adenosine monophosphate) concentration closes basolateral K+ channels . Different types of sweeteners have different chemical reactions . The neurotransmitter sends a signal to the brain of what the particular flavor is |
Science of Color |
. The segment of the electromagnetic spectrum that is visible to the human eye
. Color comes in waves of visible light . The human eye can process wavelengths from 380 (violet) to 700 (red) nanometers . When an atom goes from an excited state to a ground state, a photon is released . The wavelength of the photon determines the color . Light receptors exist in eyes, which transit photon messages to the brain to be observed as color . When light is dispersed through a prism, it splits into the colors of the visible light spectrum . The spectrum doesn’t include every color humans can see, some are unsaturated colors which mix wavelengths from multiple different colors Color Theory . A scientific and artistic theory dictating how to use color for a desired effect . Colors are organized on the color wheel . Further divided into primary, secondary, and tertiary colors . Designed by Sir Isaac Newton . Factors that affect individual color: hue, tint, shade, and tone . Color Schemes: . Complementary: 2 colors together on opposite sides of the wheel . Analogous: 3 colors side-by-side .Triadic: 3 colors spaced equally on the wheel . How we used Color Theory: . Complementary color scheme used with brown of the base of the cake and red of the marzipan . Considered all the above factors in the color of marzipan and cake |
Reflection
One of the peaks of this project was that I expanded my collaboration skills. I learned more about how to work with a group of people, both at school and outside of it. Baking this cake was a team effort and we all had to learn where we could contribute. We had to navigate around conflicts and disagreements, while remaining civil and productive. The other peak of this project was that we were able to express our creativity. We designed a cake based on a game that we had all played before, and found a creative way to tie it into our chemistry project. It was interesting to learn about all the different ways chemistry is used in our daily lives, without us even realizing.
One of the negative aspects of this project was that I struggled to be a conscientious learner. With such a lively group of people, I was always surrounded by numerous distractions, and this made staying on task difficult. I often found my concentration wondering, and I wasn't always focused on the task at hand. Another negative aspect was the lack of organization. We often found ourselves without key supplies that we needed to do our project. For example, when we were baking the cake the first time, we forgot a sifter, powdered sugar, and frosting. We had to make numerous runs to the grocery store to pick up supplies, which made the cake baking process take much longer.
One of the negative aspects of this project was that I struggled to be a conscientious learner. With such a lively group of people, I was always surrounded by numerous distractions, and this made staying on task difficult. I often found my concentration wondering, and I wasn't always focused on the task at hand. Another negative aspect was the lack of organization. We often found ourselves without key supplies that we needed to do our project. For example, when we were baking the cake the first time, we forgot a sifter, powdered sugar, and frosting. We had to make numerous runs to the grocery store to pick up supplies, which made the cake baking process take much longer.